Breakthrough Ideas for Parkinson’s Disease Treatment
MedicationParkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by bilateral motor (eg, bradykinesia, resting tremor, postural instability, rigidity) and nonmotor (eg, memory loss, depression) disability.
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by bilateral motor (eg, bradykinesia, resting tremor, postural instability, rigidity) and nonmotor (eg, memory loss, depression) disability. About 10 million people worldwide suffer from Parkinson’s disease. Current PD treatments are limited to motor symptom management, such as dopamine replacement therapy or enhancing the activity of remaining dopaminergic neurons. There are no known treatments that slow progression or prevent the onset of the disease. Furthermore, PD patients are growing at an unprecedented rate and are expected to increase to 17.5 million by 2040. Although aging remains a major risk factor for PD, more than 20 genes have been found to be associated with PD onset and progression, suggesting therapeutic potential for Parkinson’s disease.
The current drugs for the treatment of Parkinson’s disease are mainly levodopa, which forms dopamine through decarboxylation in the brain, combined drugs, and monoamine oxidase B (MAO-B) inhibitors. Due to their unsatisfactory clinical therapeutic effects, new targets are clinically needed for the treatment of Parkinson’s disease. Existing studies have found that leucine-rich repeat kinase 2 (LRRK2) mutation is the most common genetic risk factor for Parkinson’s disease.
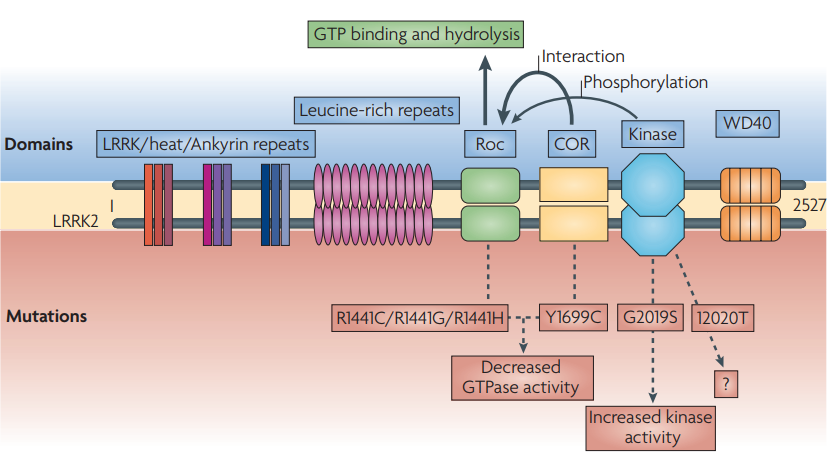
LRRK2 is a large protein consisting of 2527 amino acids including multiple domains. It contains kinase and GTPase domains, and some protein-protein interaction domains, such as: ankyrin (ANK), leucine-rich repeat (LRR), ROC and WD40. Increased LRRK2 kinase activity is thought to impair lysosomal function and may contribute to Parkinson’s disease, the study found. LRRK2 mutations account for 4-5% of familial Parkinson’s disease and 1-2% of sporadic Parkinson’s disease. Inhibition of LRRK2 may therefore be a promising disease-modifying therapeutic target in Parkinson’s disease.
Specific LRRK2 mutations increase kinase activity, leading to phosphorylation of Rab GTPases, which in turn leads to lysosomal dysfunction and Parkinson’s disease-associated pathology. Increased LRRK2 activity and pRab independent of LRRK2 mutation status were also observed in Parkinson’s disease patients. In the Parkinson’s disease model, inhibiting LRRK2 can restore the function of lysosomes, and it is effective for patients with and without LRRK2 gene mutations.
Mutations in LRRK2 mainly occur in the GTPase and kinase domains, resulting in increased kinase activity and autophosphorylation. To date, a large number of disease-associated LRRK2 mutations have been identified, among which five missense mutations (R1441C, R1441G, Y1699C, G2019S, and I2020T) have been implicated in PD pathogenesis. There have been extensive drug discovery efforts to develop LRRK2 inhibitors, particularly ATP-competitive active-site kinase inhibitors.
Figure 1. Domains and mutations of LRRK2.
Currently reported LRRK2 kinase inhibitors include LRRK2-IN-1, CZC-25146, TAE684, GNE-7915, HG-10-102-01, MLi-2, PF-06447475, DNL201 and DNL151, etc., among which DNL201 and DNL151 have entered clinical practice. LRRK2-IN-1 is the first reported selective inhibitor of LRRK2 kinase.
These inhibitors are all ATP-competitive type 1 kinase inhibitors that bind to a conformation that closes active LRRK2. However, these inhibitors lead to dephosphorylation of Ser935 causing adverse effects in treated patients, such as interference with cellular vesicle trafficking, and may lead to undesirable on-target side effects observed in the lung and kidney. Therefore, there is a need to find new strategies to replace LRRK2 targeting. PROTAC technology is a new drug research and development technology that has been very hot in recent years. It consists of three parts, one part is the target protein inhibitor, the other part is the E3 ligase inhibitor, and the middle part is connected by different linking chains. Compared with traditional small molecule inhibitors, it has the advantages of being able to target undruggable targets, high selectivity, and high activity. PROTACs are not just an emerging drug discovery paradigm, but also provide new chemical tools for target identification and validation and deciphering target biology. Through PROTAC technology, the scientists synthesized a complex-XL01126 based on the LRRK2 inhibitor HG-10-102-01 linked to different E3 ligase ligands (including VHL, CRBN and cIAP). Through its optimization and evaluation, it was found that XL01126 serves as a suitable degrader probe for studying the non-catalytic and scaffolding functions of LRRK2 in vitro and in vivo, and provides an attractive starting point for future drug development.
As aging becomes more and more serious, more and more patients with Parkinson’s disease are attracting more and more people’s attention. At present, there are no good drugs for the treatment of Parkinson’s disease on the market, so LRRK2 inhibitors and PROTACs are developed to the treatment of Parkinson’s disease is worthy of in-depth research, and it is believed that there will be milestone breakthroughs in the near future.